 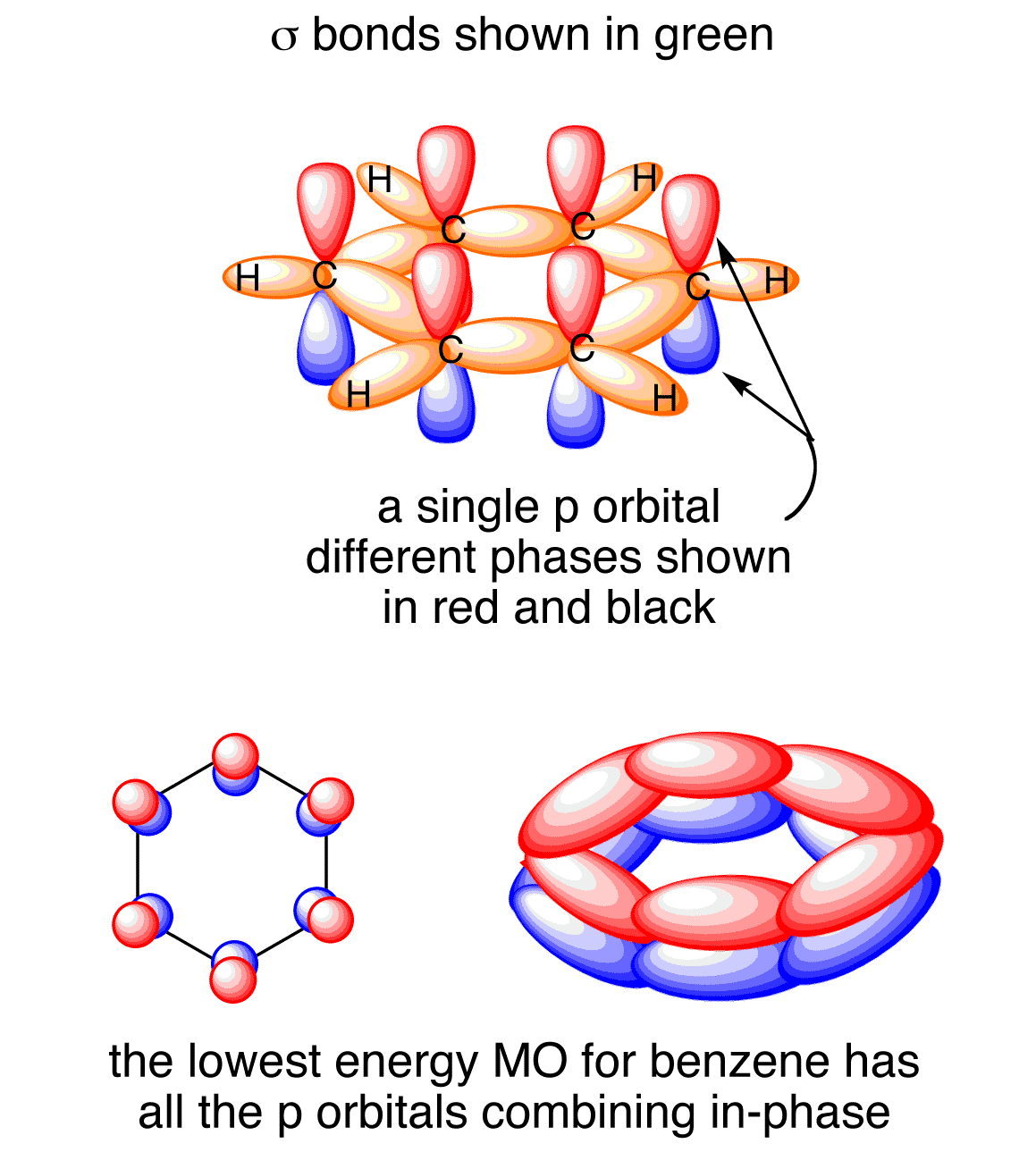
Necessary cookies are absolutely essential for the website to function properly. There are 6 delocalised electrons in Benzene. The presence of delocalised electrons is a hallmark of aromatic compounds. Which of the following has delocalized electrons? 
The localised $pi$ orbital is less stable than the delocalisation $pi$ orbital. The whole molecule can be delocalised by pairs of bonding electrons extending over three or more atoms. What do you mean by delocalization Class 11? The difference between resonance and delocalization is that resonance refers to the stabilization of a molecule due to delocalization of electrons, while delocalization refers to the distribution of electrons throughout a molecule. What is the difference between resonance and delocalization? The electrons can be delocalized all the way around the ring, above and below the plane of the ring, thanks to Benzene’s sp2 hybridized ring atoms. They can’t be in contact with each other. The p-orbitals of the carbon atom are not delocalized by CO2. electrons are still contained in the atoms, but not on the valent shell Is CO2 delocalized? When they go through metallic bonding, the electrons on the valent shell become delocalized. See also What Rarity Are Tw Zombas? Are all electrons delocalized in metals? The outer electrons form a pool of delocalized electrons that are free and move through the lattice of positive ion atoms. In the model of theelectron sea, atoms in a metallic solid lose their electrons and form a lattice of positive metallic ion. The core of the atom is exposed when there is a transfer of electrons from one group to another. Transferring electrons from one atom to another is called stems. Fourteen of the 18 electrons from the three oxygens are used for this, and four of them are used for pi bonding. electrons can occupy orbitals belonging to the molecule as a whole, which is why the same molecule can be handled by the same theory. How many delocalized electrons are in Ozone? The electrons forming the bond between the linked atoms stay between them. Delocalized electrons bring extra stability to a system compared to a similar system where electrons are not delocalized. The stabilizing force of charge delocalization is that it spreads energy over a larger area than if it were confined to a small area. Why are delocalized electrons more stable? The -electrons are delocalized as shown in the diagram below. The electrons in the p orbitals are referred to as -electrons. Strong metallic bonding occurs when delocalised electrons are shared. The electrons from the outer shells of the metal atoms can move through the whole structure. See also What Are The Two Capitals Of Sri Lanka? Why are electrons delocalized in metals?īig structures of atoms are found in metals. The delocalisation of electrons is shown by a circle. A delocalised bonding electron is an electron that is not associated with a single atom or a covalent bond. Localized bonds are described as this type of bond. What are Localised and delocalized bond give example? When atomic orbitals overlap, the strength of a covalent bond is proportional to the amount of overlap, which is assumed to be in the local bonding model. What does localized mean in chemistry?Ī specific bond or lone pair on a specific atom is one of the types of localizedmolecular orbitals. What is the meaning of delocalized electron?ĭelocalized electrons are electrons that are not associated with a single atom or a covalent bond. What is the difference between delocalized and localized?ĭelocalised chemical bonds are a specific bond that is not associated with a single atom or a covalent bond. Which of the following has delocalized electrons?.What do you mean by delocalization Class 11?.What is the difference between resonance and delocalization?.Does benzene have delocalised electrons?.Are all electrons delocalized in metals?.How many delocalized electrons are in Ozone?.Why are delocalized electrons more stable?.Are delocalized electrons in p orbitals?.Why are electrons delocalized in metals?.What are Localised and delocalized bond give example?.What is the meaning of delocalized electron?.What is the difference between delocalized and localized?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
